 Allergic rhinitis and hay fever
Allergic rhinitis and hay fever
-
1. Allergic rhinitis and hay fever
- 1.1 Why does allergic rhinitis matter?
- 1.2 Definitions
- 1.3 Diagnosis
- 1.4 Skin prick and RAST (IgE) testing
- 1.5 Allergen avoidance
- 1.6 When to start treatment in seasonal rhinitis?
- 1.7 Management options: non-pregnant adults ≥12y
- 1.8 Management options: pregnancy, breastfeeding and children <12y
- 1.9 Summary of available drugs
- 1.10 Advice for people buying over the counter
- 1.11 Antihistamines
- 1.12 Intranasal corticosteroid sprays
- 1.13 Combined intranasal corticosteroid and intranasal antihistamine: the evidence
- 1.14 Decongestants
- 1.15 Persistent eye symptoms
- 1.16 Intramuscular depot steroids
- 1.17 When to refer
- 1.18 Allergen immunotherapy
- 1.19 Intranasal phototherapy
- 1.20 Cryotherapy for chronic rhinitis (allergic and non-allergic)
- 1.21 Protective factors
- 1.22 Treatment failure: differential diagnosis
This Pearl is provided as free content. Here is the link to our terms of use
Allergic rhinitis and hay fever
Allergic rhinitis and hay fever
This article considers all types of allergic rhinitis, including hay fever. There is growing prevalence of hay fever in adults and children, thought to be due, in part, to climate change and longer pollen seasons. We may see it as something trivial – but, in fact, it has a significant impact on quality of life and sleep, and can impact exam results.
Would we feel differently if we thought of it as ‘asthma of the nose’?
In addition, there may be inequitable access to treatment because many areas in England have asked practices to signpost people to self-care with over-the-counter treatments. This is often appropriate, and we can signpost people to shop around for the cheapest generic formulations. But, in the current economic climate, we need to be mindful of the impact of these blanket rules on more socially-deprived (CORE20+5) populations, and the potential impact of untreated moderate and severe hay fever on work and school.
This article was reviewed and updated in November 2025.
It considers all types of allergic rhinitis, including hay fever. It is based on:
- The British Society for Allergy and Clinical Immunology (BSACI) guideline for the management of allergic and non-allergic rhinitis (NICE accredited) (Clin Exp Allergy 2017; 47:856).
- International consensus statement on allergy and rhinology: Allergic rhinitis 2023 (ICAR 2023;13(4):293) – a comprehensive systematic review.
Other sources are referenced where relevant.
Why does allergic rhinitis matter?
The BSACI guideline (Clin Exp Allergy 2017; 47:856) reminds us that allergic rhinitis is important because it:
- Is common, affecting 10–15% of children and 26% of adults in the UK (for some, it may go into remission as an adult).
- Impacts on quality of life, including:
- In those taking exams: symptomatic seasonal allergic rhinitis has been associated with a negative impact on exam results (data from the UK, J Allergy Clin Immunol 2007;120(2):381).
- Sleep: can have significant negative impact on sleep (ICAR allergic rhinitis 2023 statement).
- Increases the risk of developing asthma. This is called the ‘allergic march’, where allergic rhinitis develops before the symptoms of atopic asthma. Adequate treatment of allergic rhinitis may prevent this (ICAR allergic rhinitis 2023 statement).
- Is common in those with asthma (74–81% report rhinitis symptoms) and increases the risk of acute asthma exacerbations.
- Is often under/sub-optimally treated – and while many treatments are now available over the counter from community pharmacy, correct use of these at the correct time can make a REALLY big difference.
Definitions
Allergic rhinitis is an IgE-mediated type 1 hypersensitivity reaction of the nasal mucus membranes that occurs when a sensitised person is exposed to an allergen.
| Seasonal allergic rhinitis | Symptoms occurring at specific times of year, typically associated with outdoor allergens. |
| Perennial allergic rhinitis | Symptoms occurring all year round, typically associated with indoor allergens. |
The ICAR allergic rhinitis 2023 statement reminds us that climate change (lengthening many of the classical pollen seasons) and sensitisation to multiple allergens can make these definitions challenging. Allergic rhinitis can therefore be seasonal, perennial or both!
Historically, guidelines talked about intermittent and persistent symptoms. However, these terms have completely disappeared from the newer guidelines – and we are secretly glad because we weren’t sure how helpful they were to us in primary care anyway!
Severity of allergic rhinitis can be measured using the ARIA (Allergic Rhinitis and its impact on Asthma) classification:
| Mild | Moderate or severe |
|
|
In reality, we think this means that most people who seek help in primary care will have moderate or severe allergic rhinitis!
Diagnosis
We can usually make a clinic diagnosis of allergic rhinitis in primary care (Clin Exp Allergy 2017; 47:856).
Take a history
This will be based on typical symptoms, some or all of which may be present:
- Nasal congestion/coryza/sneezing.
- Nasal blockage (if unilateral, consider other possibilities; if bilateral, may indicate polyps or septal deviation – in either case, look up the nose!).
- Sinus symptoms.
- Itchy/inflamed/watery/swollen eyes.
- Itchy mouth or palate.
- Cough, wheeze, shortness of breath (consider co-diagnosis of asthma and treat if present).
Pollen food syndrome may also be present – see Pollen food syndrome (oral allergy syndrome) in the online handbook.
Ask about family history of atopy; this adds weight to the diagnosis, but its presence is not essential.
What are the triggers?
We can ask when and in what circumstances the person experiences symptoms. This can identify the likely trigger(s) and whether this is seasonal or year-round.
|
Pollen
|
This is the most likely culprit if there are seasonal symptoms. The concept of seasonal vs. perennial (year-round) symptoms can be useful in countries such as the UK where there are distinct pollen seasons. These vary in timing and intensity from year to year, but, in general:
|
| House dust mite |
Consider if year-round symptoms, worse on waking and experienced indoors. While symptoms may be year-round, they may peak in spring and autumn. |
| Animal dander | Symptoms following exposure to affecting animal(s). |
| Occupational | If symptoms are more severe at work and improve during weekends/holidays, consider an occupational trigger. Typical triggers may include latex, chlorine, flour, wood dust, laboratory animals. |
Examination
Examination may be normal in someone with allergic rhinitis.
We may see ‘allergic shiners’ (dark discolouration of lower eyelid), periorbital oedema or throat clearing.
Looking up the nose may be helpful to identify typical allergy findings, e.g. inflamed hypertrophied turbinates, rhinorrhoea, and to identify things we might not expect, which will need separate treatment.
If nasal blockage predominates, check for polyps (these are greyish blue in colour and not sensitive to touch, unlike inflamed turbinates which are pink and very sensitive!).
Skin prick and RAST (IgE) testing
The BSACI guideline states that “skin prick testing should be routinely carried out to determine if rhinitis is allergic or non-allergic and interpreted considering the clinical history” (Clin Exp Allergy 2017; 47:856).
At Red Whale, we do not think this is routine practice for most people presenting to primary care in whom we make clinical diagnosis (or those self-diagnosing hay fever symptoms).
Skin prick testing may be useful where we suspect an allergen trigger that may be avoidable, e.g. animal dander or occupational exposure. Access is variable in UK primary care, and many of us would likely use advice and guidance or refer.
The ICAR consensus statement on allergy and rhinology 2023 states that clinical diagnosis of allergic rhinitis is reasonable (ICAR 2023;13(4):293).
It states that diagnostic testing is useful in the following situations:
- Immunotherapy is being considered.
- Targeted allergen avoidance is being considered, e.g. animal dander or occupational exposure.
- Inadequate response to treatment.
It also reminds us that many people who appear sensitised to allergens on skin prick will not demonstrate any allergy symptoms. It is therefore important to correlate test findings with the clinical picture (we may meet this in individuals who have done online or private allergy testing).
Access is variable in UK primary care and many of us would likely use advice and guidance or refer.
Remind me about skin prick and RAST (IgE) testing…
There are two types of test that can be used to identify type 1 hypersensitivity reactions. Skin prick is considered gold standard, with RAST (IgE) testing an option for those who cannot have skin prick testing. Here is a brief description of the two tests (Malays Fam Physician 2021;16(2):19):
| Skin prick testing |
|
| RAST (IgE) testing |
|
Allergen avoidance
Allergen avoidance isn’t always easy or possible.
The BSACI guideline acknowledges this, and reminds us that the evidence is mostly low grade. It suggests that we give the following information (Clin Exp Allergy 2017; 47:856):
| Pollen |
|
| House dust mite |
A 2012 Cochrane review suggested:
|
| Pet allergens |
|
The BSACI guideline also notes that air/traffic pollution and cigarette smoke exposure can be exacerbating factors and should be avoided.
Let’s consider management…
When to start treatment in seasonal rhinitis?
For seasonal rhinitis, a key message is to start treatment at least 2 weeks before usual symptoms start. This pollen calendar is adapted from UK Met Office data (Primary Care Respiratory Update 2022 Issue 24).
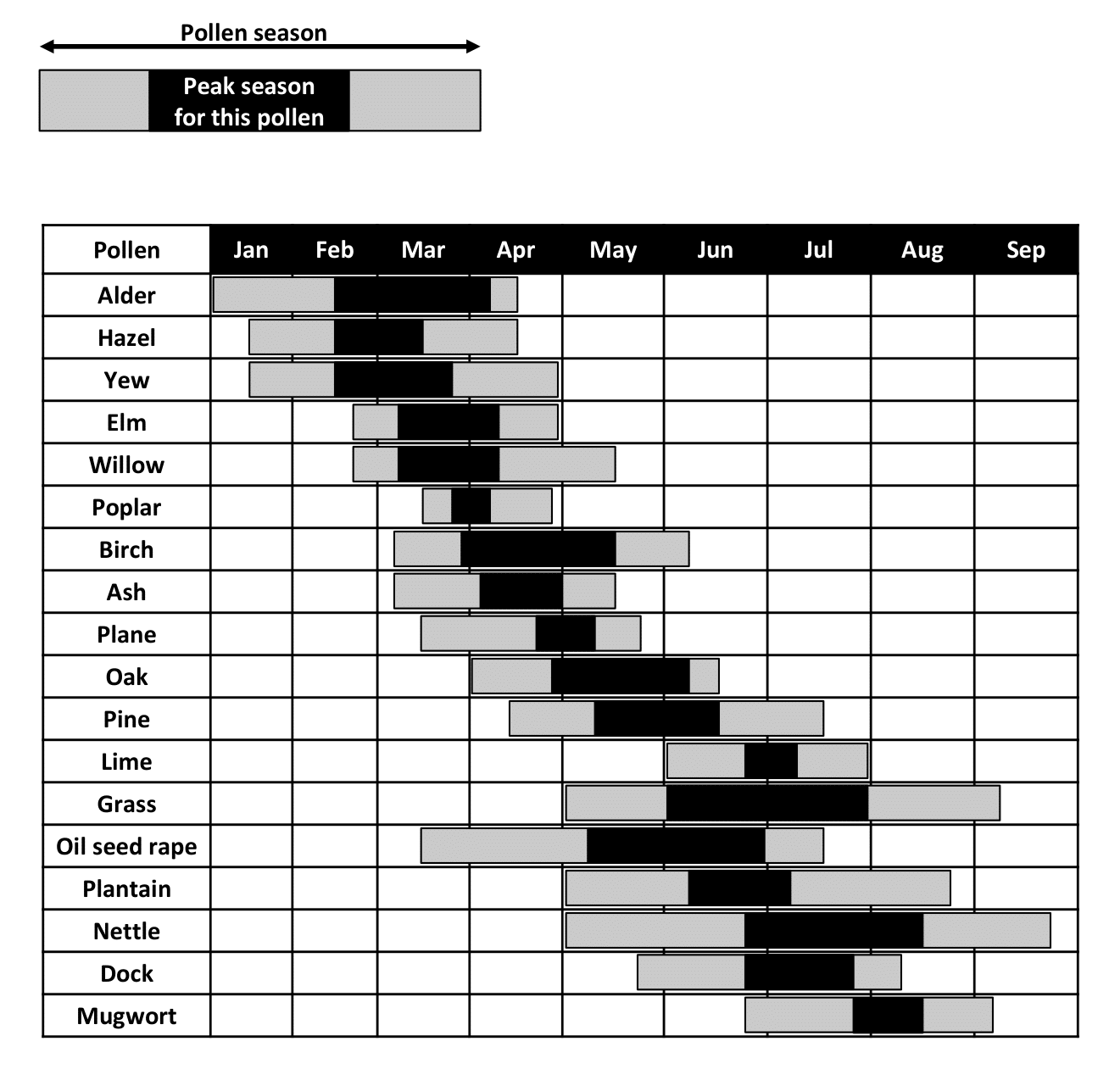
Practical tip: If someone knows which pollen(s) they are allergic to, they might find a pollen calendar useful as a reminder (see link in useful resources, below). They could then be encouraged to set a reminder, e.g. on their phone, to start treatment. The Primary Care Respiratory Society suggests that for people with asthma and seasonal allergic rhinitis, we could consider setting their medication review a few weeks before their typical ‘allergy season’ to reinforce this message (Primary Care Respiratory Update 2022 Issue 24).
Management options: non-pregnant adults ≥12y
Please follow the link for a PDF version of the GEMS for download/printing: Allergic rhinitis and hay fever: GEMS
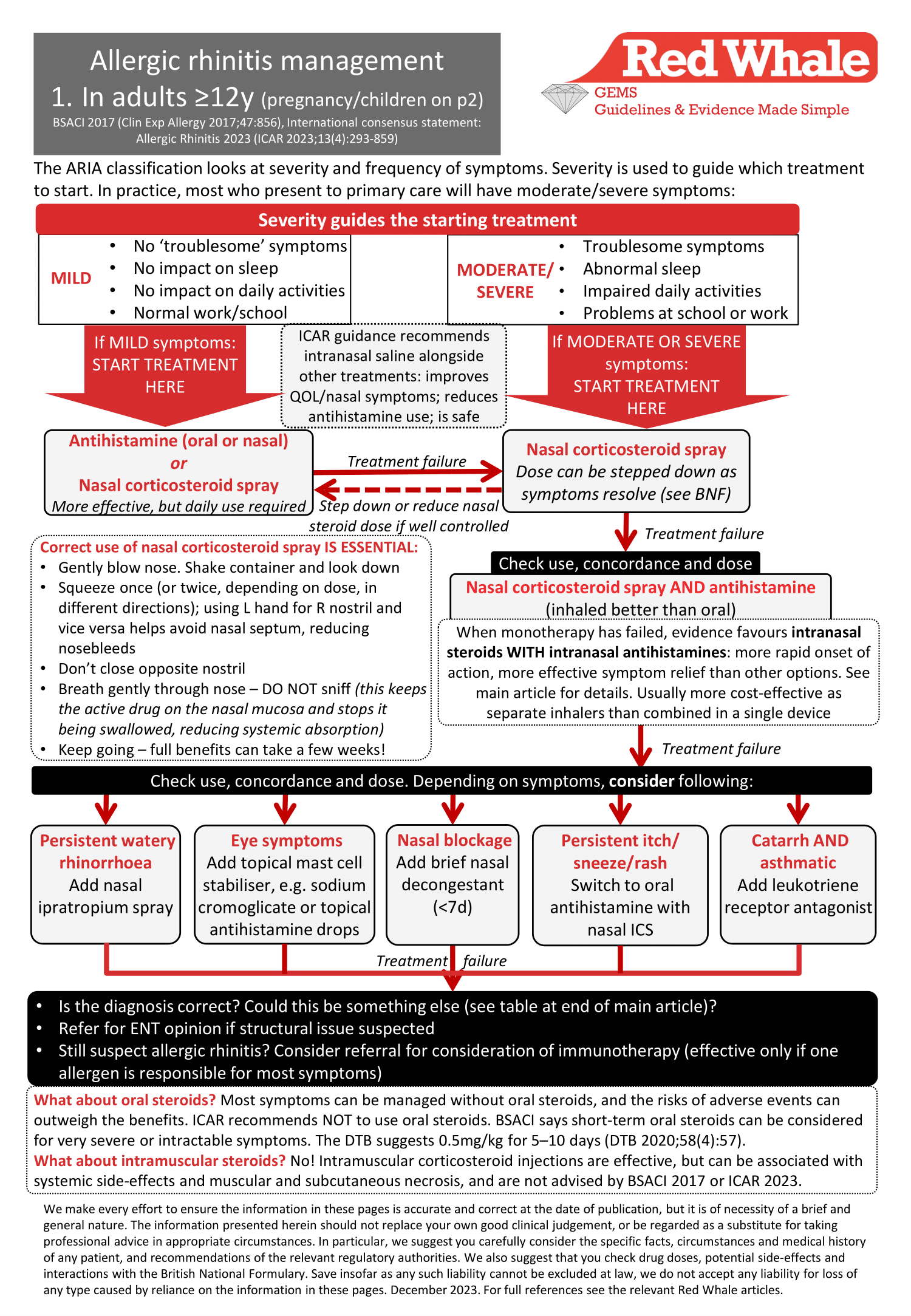
(BSACI guideline for the diagnosis and management of allergic and non-allergic rhinitis (Clin Exp Allergy 2017;47:856), International Consensus Statement: Allergic Rhinitis 2023 (ICAR 2023;13(4):293-859), DTB 2020;58(4):57)
Management options: pregnancy, breastfeeding and children <12y
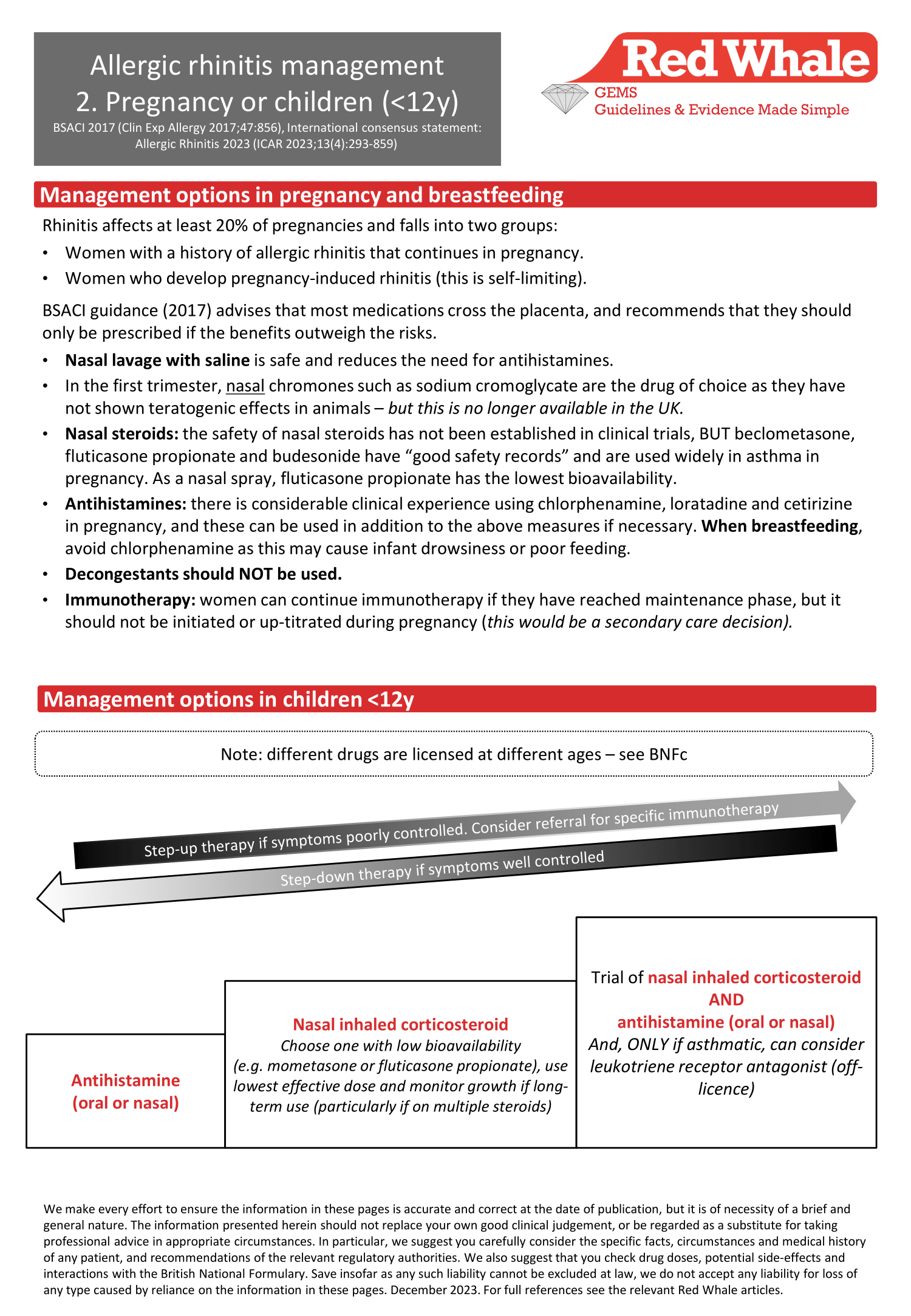
(BSACI guideline for the diagnosis and management of allergic and non-allergic rhinitis (Clin Exp Allergy 2017;47:856), International Consensus Statement: Allergic Rhinitis 2023 (ICAR 2023;13(4):293-859), DTB 2020;58(4):57)
Summary of available drugs
This table shows the current commonly-available core drugs in the UK. It lists those which are prescription only and those that are also available over the counter. For doses, see the BNF and BNFc.
| Drug | OTC or POM | Licensed in children? |
| Antihistamines (only newer generation listed) | ||
| Cetirizine (oral) | OTC | Not licensed for use age <2y |
| Loratadine (oral) | OTC | Not licensed for use age <2y |
|
Fexofenadine (oral)
|
OTC
|
Not licensed for use age <6y (Note: 180mg dose not licensed for hay fever) |
| Acrivastine (oral) | OTC | Not licensed for use age <12y |
| Rupatadine (oral) | POM | Not licensed for use age <12y |
| Desloratadine (oral) | POM | Not licensed for use age <1y |
| Azelastine (nasal spray) | POM |
Not licensed for use age <6y
|
| Nasal inhaled corticosteroids | ||
|
Beclometasone (not preferred by BSACI because of higher bioavailability) |
OTC |
Not licensed for use age <6y
|
| Mometasone | OTC |
Not licensed for use age <3y
|
| Fluticasone propionate | OTC |
Not licensed for use age <4y
|
|
Fluticasone furoate (Avamys) NOTE: double the potency of fluticasone propionate |
POM |
Not licensed for use age <6y
|
| Combined nasal steroid and antihistamine (note: some local prescribing guidelines will not allow prescription of the combined form, either at all or until separate inhaled nasal steroids and inhaled nasal antihistamines have been tried) | ||
|
Fluticasone propionate with azelastine (nasal spray) (Dymista) |
POM OTC (as Dymista Control) |
Not licensed on prescription for use age <12y Not licensed OTC for use age <18y |
|
Mometasone furoate with olopatadine (nasal spray) (Ryaltris) |
POM | Not licensed for use age <12y |
Now, let’s consider the evidence and some frequently asked questions…
Advice for people buying over the counter
Many areas have asked general practice not to prescribe for hay fever and to instead recommend over-the-counter medication. However, we recognise that, for some, this can be difficult, and it has become such a hot topic that even Money Saving Expert has a ‘best buys’ for hay fever page. We can advise patients to:
- Buy the cheapest preparation (often unbranded) that contains the active ingredient of the preparation they use and find effective.
- Whether branded or generic, all medicines are made to the same standard.
There are useful links to price comparison sites in the useful resources box, below.
While, for most, over-the-counter treatment is completely appropriate (particularly with good advice about when to start and how to use), a minority of people may need prescription-only medication to manage their symptoms – and, in the current economic climate, we may need to be mindful of the disproportionate impact on the most socially-deprived members of our communities if we have to adhere rigidly to blanket rules.
Antihistamines
BSACI (2017) and ICAR (2023) recommend oral or intranasal antihistamines as a first-line treatment option for all types of allergic rhinitis where symptoms or impact are mild. The other option is inhaled nasal steroids (but these must be used daily to get benefit, and some people with mild or intermittent symptoms do not want to use something daily).
Antihistamines should be taken regularly in persistent rhinitis, but can be used as-needed for intermittent symptoms.
Oral antihistamines
- Reduce symptoms by, on average, 7% compared with placebo.
- Can improve quality of life for people with hay fever and allergic rhinitis.
Which should we choose?
Do not use first-generation antihistamines, e.g. chlorphenamine, diphenhydramine or promethazine.
Instead, choose newer-generation antihistamines (e.g. cetirizine, loratadine – see table above for all currently available options). They are also recommended in combination with inhaled nasal steroids for those with moderate or severe symptoms who do not get sufficient relief with inhaled nasal steroids alone.
Why should we not use first-generation antihistamines?
First-generation antihistamines, e.g. chlorphenamine, diphenhydramine, promethazine, are not recommended for use in allergic rhinitis because:
- They are less efficacious than second and third-generation antihistamines.
- They cross the blood–brain barrier, and can cause drowsiness and impair driving. One study with diphenhydramine showed that it impaired driving more than being over the legal limit for alcohol, and self-reported drowsiness ratings were not a good predictor of impairment (Ann Int Med 2000;132(5):354).
- They may also have an anticholinergic effect, which has been associated with an increased risk of dementia.
Intranasal antihistamines
- Intranasal antihistamines have been shown to be equal or superior to oral antihistamines in symptom improvement, and tend to have a more rapid onset of action.
- They are more effective than nasal corticosteroids for eye symptoms.
- Only azelastine nasal spray is currently available as a monotreatment in the UK, and it is a prescription-only medication.
Intranasal corticosteroid sprays
ICAR 2023 reminds us that:
- These are considered first-line therapy for allergic rhinitis for those with moderate or severe symptoms (most people we will see in primary care), and are an option for first-line treatment for those with mild symptoms.
- Inhaled nasal steroids are superior for nasal and ocular symptoms when compared with oral antihistamines and leukotriene receptor antagonists.
- Some benefits may be seen after 8 hours, but maximal benefits may take up to 2 weeks. For seasonal rhinitis (hay fever), should ideally be started 2 weeks before ‘usual’ symptoms would start.
- Children should take the lowest effective dose of a low-bioavailability preparation.
IMPORTANT: Using an inhaled corticosteroid spray correctly makes a BIG impact on how effective it is – see the GEMS for details of how to explain this.
Which nasal corticosteroid?
All available corticosteroid nasal sprays have similar efficacy. This means that patient preference (including smell and aftertaste!), number of times per day dosing and ease of use of device may all be relevant.
The bioavailability of inhaled nasal corticosteroids varies significantly, and this is of relevance in children and those who may be taking other steroids, e.g. asthmatics, particularly when long-term use is considered.
BSACI (2017) reminds us that:
- Systemic absorption is minimal for fluticasone propionate, fluticasone furoate and mometasone furoate. For this reason, these preparations are preferred in children and for long-term use.
- Systemic absorption of other preparations varies:
- Budesonide 33%.
- Beclometasone dipropionate 44%.
- Triamcinolone 44%.
- Betnesol 100%.
Combined intranasal corticosteroid and intranasal antihistamine: the evidence
ICAR 2023 makes a strong recommendation for a combination of intranasal corticosteroid and intranasal antihistamine when monotherapy has failed to control symptoms.
Grade A evidence now shows that a combination of intranasal corticosteroid spray and intranasal antihistamine has a more rapid onset of action and more effective symptom relief than either intranasal corticosteroids or intranasal antihistamine alone, and that taking both drugs nasally is more effective than intranasal corticosteroids with oral antihistamine. ICAR 2023 comments that concurrent use of two separate nasal sprays may be more cost-effective than a combined preparation.
What are your local prescribing guidelines?
Decongestants
ICAR 2023 makes a strong recommendation against routine use of decongestants, but states that they can be used for short-term (<7d) ‘rescue therapy’ for nasal congestion not responding to other management options.
Routine use, particularly of nasal decongestants, increases the risk of rhinitis medicamentosa (see table at the end of this article).
Persistent eye symptoms
BSACI 2017 reminds us that, for many people, allergic rhinitis is, in reality, allergic rhinoconjunctivitis. For most people, managing the rhinitis with inhaled nasal corticosteroids +/- nasal antihistamines will manage eye symptoms. However, some will need topical eye drops. These can be used in addition to other treatments, and options include:
- Mast cell stabilisers, e.g. sodium cromoglycate, nedocromil sodium; these work best before symptoms develop.
- Antihistamine eye drops, e.g. azelastine.
- Olopatadine drops: have both mast-cell stabilising and antihistamine properties.
If the person is still struggling despite these measures, topical steroid drops can be used, but this would be a secondary care decision because the potential risk of glaucoma requires ocular monitoring, which we cannot do in primary care.
Intramuscular depot steroids
“Please can I have a steroid injection? That bloke on the radio says he has one every year.”
Intramuscular corticosteroid injections are effective, but can be associated with systemic side-effects and muscular and subcutaneous necrosis, and are not advised by BSACI 2017 or ICAR 2023.
The DTB reviewed the evidence in 2020 and reiterated this advice (DTB 2020;58(4):57).
What are the risks?
- Adrenal suppression, over and above a short course of oral corticosteroids.
- BSACI also cites a Danish National Registry retrospective study that demonstrated that people receiving one (or more) corticosteroid injections per year for at least 3 years had an increased risk of osteoporosis and diabetes.
When to refer
If symptoms remain persistent or severe, despite working through all the primary care therapy options, we could consider referral to specialist allergy services. They may consider allergen immunotherapy, but are likely to ensure all the basics are optimised first.
If there is significant nasal obstruction or structural abnormalities, e.g. septum deviation or persistent turbinate hypertrophy, ENT referral may be helpful.
Allergen immunotherapy
BSACI (2017) and ICAR (2023) remind us that allergen immunotherapy (AIT) is an option for people with severe refractory symptoms despite optimal pharmacological management. It has the potential to initiate ‘sustained immunological alterations’.
It works best where a person is sensitised to a single allergen, e.g. grass pollen, house dust mite, short ragweed, some tree pollen (it is not available for all allergens).
Treatment is prolonged (usually 3+ years), but can sometimes result in remission and may prevent progression to asthma in children with seasonal rhinitis. It can be delivered sublingually or subcutaneously. There is an absence of evidence for its use in children aged <5y.
House dust mite
NICE has approved the use of the immunotherapy ‘house dust mite sublingual lyophilisate’ (12 SQ-HDM SLIT, brand name Acarizax) (NICE 2025, TA1045):
- It is approved for use in people aged 12–65y with proven house dust mite allergic rhinitis who have not responded to optimal pharmacological management.
- It should be initiated in secondary care, but NICE approved continued prescribing in primary care.
In its analysis, the committee drew on research showing that 12 SQ-HDM SLIT may:
- Reduce symptoms by around 16% when compared with placebo (although evidence around the size of the benefit was uncertain).
- Improve quality of life in people with house dust mite allergic rhinitis.
- Reduce the use of other prescription medication for rhinitis (in particular the corticosteroid burden).
Birch pollen
NICE has approved the use of betula verrucosa sublingual lyophilisate (12 SQ-Bet), brand name Itulazax) (NICE 2025, TA1087):
- It is approved for use in adults with moderate to severe allergic rhinitis and/or conjunctivitis caused by pollen from the birch tree family (Including birch, alder, hornbeam, oak, beech and hazel), as an add-on to usual care.
- Patients must have proven allergy on skin test or allergen-specific IgE, and persistent symptoms despite usual treatments.
- Phase 3 clinical trials showed a 47% reduction in rescue-medication use and a 33% relative reduction in symptom severity scores when compared with placebo (in combination with usual treatments) (12SQ-Bet SMPC accessed November 2025).
The BNF reminds us that this drug must be initiated in secondary care (BNF accessed November 2025). Treatment should be started at least 4 months prior the start of the pollen season and be continued throughout the season, repeated annually for 3 years.
Intranasal phototherapy
You may have seen (or own) one of the devices with light probes that poke up the nose and deliver intranasal phototherapy. I have had several patients ‘swear’ by them.
There are both self-administered devices which can be purchased at community pharmacies, and other devices that can be administered by a clinician. Intranasal phototherapy is claimed to increase local blood flow and suppress inflammation.
NICE recently reviewed the evidence, and concluded that evidence on efficacy and safety was limited, so it should only be initiated by clinicians in the context of a clinical trial (NICE 2018; IPG 616). Of course, patients may still purchase their own device!
Cryotherapy for chronic rhinitis (allergic and non-allergic)
NICE has reviewed the evidence for cryotherapy to the posterior nasal nerve to treat chronic rhinitis, and found there was insufficient evidence to recommend use outside of trials (NICE 2023 IPG771). The committee noted that short-term evidence is promising, but it remains uncertain which patients will benefit most and how long the effect of treatment would last.
Protective factors
“Is there anything I can do to stop my kids getting allergic rhinitis as badly as me?”
ICAR 2023 reminds us that there is still a mixed evidence base here and that further research is needed. Based on the currently available studies:
- Breastfeeding: mixed evidence. There is a slight tendency to benefit over harm for protection against allergic rhinitis. The authors concluded that it should be recommended because it is low cost, has no associated harms and has wider benefits for the general health of infants.
- Childhood exposure to pets: conflicting evidence. In ‘non-allergic families’, early pet exposure in the first year of life, particularly to dogs, may be protective. No specific recommendation was made based on absence of evidence.
Treatment failure: differential diagnosis
All that sneezes and has a runny nose is not allergic rhinitis. If we see someone who is not responding as we expect, despite good concordance with treatment, we should consider alternatives or factors which may be exacerbating the underlying condition. The differential is broad and includes (ICAR 2023):
| Conditions | Features/management |
| Rhinosinusitis (chronic) |
|
| Drug-induced rhinitis |
|
| Rhinitis medicamentosa |
|
| Occupational rhinitis |
|
| Smoke-induced rhinitis |
|
| Infectious rhinitis (URTI) |
|
| Hormonally-induced rhinitis |
|
| Pregnancy-related rhinitis |
|
| Non-allergic rhinitis with eosinophilia syndrome (NARES) |
|
| Non-allergic rhinopathy and vasomotor rhinitis |
|
| Autoimmune, granulomatous and vasculitic rhinitis |
|
In practice, in primary care, common things will be common; for many of these conditions, the mainstay of first-line management is, you guessed it, inhaled nasal steroids. But, in some of these scenarios, allergy testing may be useful. We will be most likely to get the right people to allergy services if we have given good explanations and demonstrations of how to use inhaled nasal corticosteroids!
|
Allergic rhinitis and hay fever
|
|
|
Useful resources for patients: Websites (all resources are hyperlinked for ease of use in Red Whale Knowledge)
|
This information is for use by clinicians for individual educational purposes, and should be used only within the context of the scope of your personal practice. It should not be shared or used for commercial purposes. If you wish to use our content for group or commercial purposes, you must contact us at sales@red-whale.co.uk to discuss licensing, otherwise you may be infringing our intellectual property rights.
Although we make reasonable efforts to update and check the information in our content is accurate at the date of publication or presentation, we make no representations, warranties or guarantees, whether express or implied, that the information in our products is accurate, complete or up to date.
This content is, of necessity, of a brief and general nature, and this should not replace your own good clinical judgment or be regarded as a substitute for taking professional advice in appropriate circumstances. In particular, check drug doses, side effects and interactions with the British National Formulary. Save insofar as any such liability cannot be excluded at law, we do not accept any liability for loss of any type caused by reliance on the information in these pages.
Here is the link to our terms of use.