 Gout
Gout
-
1. Gout
- 1.1 Gout basics
- 1.2 Why is gout important?
- 1.3 Gout: NICE 2022
- 1.4 Diagnosis
- 1.5 Imaging
- 1.6 Managing acute attacks
- 1.7 Offer follow-up to all after the first acute attack
- 1.8 Diet
- 1.9 Urate-lowering therapy: deciding whether to start
- 1.10 Treat-to-target approach
- 1.11 Choice of urate-lowering therapy agent
- 1.12 Febuxostat and cardiovascular disease
- 1.13 Drug doses
- 1.14 Doses of allopurinol in impaired renal function
- 1.15 Antihypertensives and gout
- 1.16 Is urate an independent risk factor for cardiovascular disease?
- 1.17 How should we organise gout care?
This Pearl is provided as free content. Here is the link to our terms of use
Gout
Gout
This article was updated in May 2023.
NICE produced its first gout guideline in 2022 (NICE 2022, NG219). This supersedes the 2017 British Society of Rheumatology (BSR) guideline (Rheumatology 2017;56:e1), and we highlight the differences below. We’ve summarised the NICE guideline in the following GEMS.
Gout basics
- Gout is the most common inflammatory arthropathy, affecting 2.5% of UK adults (An Rheum Dis 2015;74:661).
- Gout is more prevalent in men (4%) than women (1%).
- Gout is caused by monosodium urate crystals which form in joints and soft tissues.
- Longer term, gout can cause joint destruction and renal stones.
- The prevalence of CKD is higher in those with gout: almost 25% of people with gout have CKD stage 3–5.
- Any joint can be affected, but it is most common in distal joints, classically the 1st metatarsophalangeal joint (MTPJ).
Why is gout important?
Because we don’t manage it as well as we could!
Despite several effective drugs being available, the management of gout in the UK has been described as “neither rational nor effective” (BMJ 2011;343:d7459).
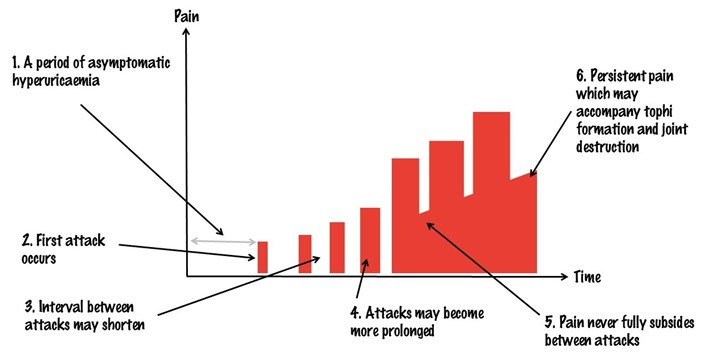
Let’s start with an important fact: gout is NOT simply a series of isolated, painful attacks of joint pain.
The figure below shows a typical natural history (BMJ 2013;347:f5648):
Chronic tophaceous gout can be associated with persistent pain, joint damage, tophi, and disability.
Gout: risk factors
High urate level has a causative relationship with gout and is the most important risk factor.
Reduced renal excretion (rather than overproduction) is responsible in 90% of cases of hyperuricaemia (Ann Intern Med 2005;143:499).
- Genetic factors are important, and most of the genes identified are involved in the renal urate-transport system. The balance of urate absorption and excretion largely determines serum uric acid level, and the effect is likely polygenetic (Nat Rev Rheumatol. 2012;8:610).
- Evidence suggests that CKD and other comorbidities, e.g. hypertension, are a risk factor for gout (BMJ Open 2015;5:e006843).
- The relationship with CKD is bidirectional, i.e. CKD is an independent risk factor for gout, but gout can predispose to CKD (Arthritis Res Ther 2018;20:243).
- Certain drugs, e.g. diuretics, low-dose aspirin and immunosuppressant agents, are a risk factor for gout (Rheumatology 2017;56:679).
- Obesity is linked with hyperuricaemia and gout (Arthritis Res Ther. 2018;20:136).
- Other lifestyle factors, including alcohol consumption, smoking and diet, are associated with increased gout risk (Lancet 2004;363:1277, Semin Arthritis Rheum 2018;47:825, NEJM 2004;350:1093).
An active lifestyle may be protective against gout (Am J Clin Nutr 2008;87:1480).
Should we treat asymptomatic hyperuricaemia?
No! NICE (NG219, 2022) and the British Society of Rheumatology (Rheumatology 2017;56:e1) both advise against treating high urate in the absence of clinical gout.
Why not? Many people with high urate never develop gout (Biomolecules 2021;11:280).
Gout: NICE 2022
Please follow the link for a PDF version of the GEMS for download/printing: Gout: GEMS

Let’s now deconstruct this guideline, ask why NICE made its decisions and compare it with the 2017 BSR guideline.
Diagnosis
Clinical suspicion
NICE keeps things ‘broad brush’ when it comes to diagnosis, offering us 3 scenarios (NICE 2022, NG219):
| Gout highly likely | Gout a possibility | Chronic tophaceous gout highly likely |
|
NICE says, ‘strongly suspect gout’:
(We think this leaves our current practice relatively unchanged: if it looks like classic gout then it probably is!)
|
NICE says, ‘consider gout’:
|
NICE says, ‘suspect chronic, untreated gout’:
|
NICE acknowledges the lack of high-quality evidence to support a straightforward, universal way of diagnosing gout in a primary care setting.
NICE has kept things very simple – and, at Red Whale, we like simple! It avoided using scoring systems such as that seen in some of the literature (Rheumatology 2015;54:609). This reflects what most of us do in practice when diagnosing gout: when it’s the big toe, it’s easy; when it’s elsewhere, it can be REALLY HARD to diagnose!
Differential diagnosis
NICE reminds us to consider the following differential diagnoses when faced with an acute inflammatory monoarthritis (NICE 2022, NG219):
- Septic arthritis (if suspected, arrange emergency assessment – likely to need joint aspiration).
-
Pseudogout (also known as calcium pyrophosphate deposition disease – see our article on Pseudogout):
- Most commonly affects the knee and wrist. Can mimic septic arthritis.
- A crystal arthropathy (calcium pyrophosphate crystals).
- Can cause calcification of the cartilage, e.g. menisci, visible on plain Xray.
- May be diagnosed on joint aspiration and microscopy.
- Other inflammatory arthritis, e.g. reactive arthropathy, psoriatic arthritis.
Blood tests: uric acid level
In its guidance, NICE:
- Notes that most people with gout have high urate (over 360micromol/l), but many people with hyperuricaemia do not have gout. In this sense, hyperuricaemia is the key risk factor for gout.
- Does not recommend treating asymptomatic hyperuricaemia (in line with other guidelines).
- Advocates checking serum uric acid (SUA) to confirm gout:
- This is a significant deviation from the British Society of Rheumatology guideline, which recommends waiting at least 4–6 weeks after the acute attack settles before checking SUA. NICE doesn’t state, but we assume this SUA check would only apply to the initial attack, rather than subsequent stereotypical attacks in someone with known gout.
- If SUA is found to be <360micromol/l, NICE suggests rechecking after at least 2 weeks as the level may be lower during a flare because uric acid is sequestered as crystals in joints.
Other blood tests
NICE does not include any other blood tests but, at Red Whale, we would suggest these additional tests may be useful if they have not been done recently; our rationale is in brackets:
- U&E (may be using NSAID or allopurinol, and CKD is more prevalent in those with gout).
- Lipids (gout is associated with CVD and metabolic syndrome, so should do a CV risk assessment).
- HbA1c (exclude diabetes: part of CV risk assessment score).
- LFT (may be using febuxostat or a statin).
Aspiration and microscopy
- NICE suggests we consider joint aspiration and microscopy (for crystals) if the diagnosis remains uncertain or unconfirmed. We should NOT be doing this to ‘rule out’ septic arthritis in primary care – if we suspect, we should refer as an emergency.
It may be the 'gold standard' method but, really…how many of us in primary care will be doing this within a 10-minute appointment?
- Not only is aspiration technically difficult, but the analysis may yield false negatives as the sample has to reach the lab in a timely manner.
- There's also the small issue of persuading the patient to let you stick a needle into their red, hot, exquisitely tender joint! That said, aspiration can be done (in rheumatology clinic) after an acute attack has settled as the crystals will often still be present in the synovial fluid.
Imaging
NICE suggests that if aspiration can't be done AND the diagnosis remains uncertain, imaging may be useful (NICE 2022, NG219). Options include:
- Plain X-ray:
- This may show changes resulting from chronic gout, e.g. erosions or tophi.
- It may show alternative or co-existing pathology, e.g. osteoarthritis.
- However, plain X-ray isn’t very sensitive in the diagnosis of acute gout (Arthritis Res Ther 2009;11:232) so we’re not sure how useful plain X-ray will be in everyday practice.
- Dual-energy CT:
- Do you have access to dual-energy CT? NICE recommends it, but acknowledges that it isn’t widely available in the UK. We’re not sure how useful this will be if we can’t access it acutely.
- Ultrasound:
- NICE concedes that it is difficult to interpret the evidence around the diagnostic utility of ultrasound.
- It included it in the guideline because it is more sensitive than plain X-ray and is “commonly used” (its words) to confirm or refute a diagnosis of gout.
We were puzzled by “commonly used”. Is NICE referring to primary care, or a secondary care setting (e.g. rheumatology clinic), or as a bedside investigation in A&E or acute assessment unit? Will your local radiology department accept requests for urgent ultrasound during an attack? In reality, ultrasound isn’t accessible for most of us in primary care so this will be a secondary care decision.
Managing acute attacks
NICE offers 3 options for treatment: NSAID, colchicine, prednisolone (NICE 2022, NG219).
NICE says that an NSAID or colchicine would normally be prescribed before corticosteroids, but any option would be reasonable based on the patient’s comorbidities, other medication and preferences. NICE advised us not to use interleukin-1 (IL-1) inhibitors as they are not cost-effective.
-
NSAIDs: NICE doesn’t specify which NSAID or indicated doses.
- In reality, most would use naproxen because of its risk/benefit profile.
- BNF recommends: 750mg, then 250mg every 8 hours until attack has passed.
- Colchicine: the BNF recommends one tablet 2–4 times a day, to a maximum of 12 tablets. Lower doses of colchicine are effective and have fewer adverse effects (BMJ 2003;327:1275).
- Prednisolone: unlicensed. A Lancet RCT used 35mg and found that this dose was as effective as naproxen over 4 days (Lancet 2008;371:1854).
What if one of these doesn’t work?
NICE suggests that if the first oral therapy is not effective, intra-articular or intra-muscular steroids can be considered. We suspect most would try a second oral agent before moving to injections!
Can you use more than one of these options at once?
- NICE makes no mention of this.
- The BSR guideline suggested that if monotherapy was insufficient, first-line therapies could be used concurrently (Rheumatology 2017;56:e1).
Is there a ‘best’ first-line choice? No.
- The evidence shows no superiority between colchicine, NSAID or oral corticosteroid. A UK head-to-head trial compared naproxen (n=200) vs. colchicine (n=199) and found no difference in pain during an acute gout flare.
- Colchicine had more adverse effects (mainly diarrhoea) but was more cost-effective.
- It concluded that, in the absence of contraindications, we should use naproxen ahead of colchicine.
- However, we note that this trial found ‘no major harms’ with naproxen, despite using a 750mg loading dose to achieve clinical parity. This is probably significantly more than you or I use in practice. We wonder if the trial lacked sufficient power to demonstrate the more significant, well-documented harms of naproxen? At Red Whale, we’re yet to be reassured that high-dose naproxen, although as effective as colchicine, is preferable to colchicine (albeit with the risk of the runs!) (Ann Rhem Dis 2020;79:276).
Offer follow-up to all after the first acute attack
Remember the graph at the beginning: we are dealing with a long-term condition, not a series of independent attacks.
- NICE recognises that, “In current practice, little or no follow-up is offered to people after a gout flare”.
Follow-up is the key step in the management pathway: without it, effective long-term management of gout is unlikely to happen. This simple step may change your practice and improve patient outcomes!
What should be included in initial follow-up?
NICE suggests:
- Check bloods, provide information, assess lifestyle and comorbidities (including cardiovascular risk):
- Why? Gout frequently co-exists with cardiovascular disease. The conditions have shared risk factors but the exact relationship between the two remains unclear. Those with coronary artery disease and pre-existing gout have worse outcomes than those without gout (JAHA 2018;7:e009328).
- How many of your patients with gout have had a QRISK? How many are taking a statin?
- Discuss the need to consider long-term management of gout, including urate-lowering therapy (see below).
- ‘Review medications’, but NICE does not go into any detail. Some medications may increase serum urate level; see our section below on ‘antihypertensives and gout’.
- Think about this pathway in your practice and/or PCN. Is this something that could be developed with the multi-professional team – perhaps one for our pharmacy and/or nursing colleagues?
Diet
NICE simply advises a ‘healthy, balanced diet’. In contrast to the BSR guideline, it does not recommend a ‘low urate diet’. It does not make recommendations to avoid ultra-processed foods, fructose-sweetened drinks or red meat, and it does not recommend cherry extract or vitamin C.
Why has NICE done this?
There is not enough evidence to show that any specific diet helps. Also, NICE recognises that other long-term conditions carry their own brand of dietary advice, raising the possibility of conflicting or contradictory messages.
At Red Whale, we’re delighted that NICE has publicised this fact: we find that complicated ‘eat these, avoid those’ lists can be a real barrier to engagement, and there is not one size fits all!
Working towards a healthier BMI, in whatever way works for that individual, and avoiding excess alcohol is likely to be the best and most straightforward advice for our patients!
Genetics probably matter more!
A meta-analysis of five cohort studies including men and women of European ancestry who had never had gout challenges the perception that diet influences serum urate level (BMJ 2018;363:k3951).
- It identified an association between some foods (alcohol and sugar-containing beverages, meat, poultry, potatoes) and elevated serum urate levels.
- But none of these foods explained even 1% of variation in urate levels.
- Genetic susceptibility had a much larger impact on high serum urate levels than variations in diet.
Urate-lowering therapy: deciding whether to start
NICE states:
| ‘OFFER’ urate-lowering therapy to those who have: | NICE suggests we 'DISCUSS' urate-lowering therapy: |
|
|
Remember: 'offer' is NICE’s strongest recommendation; ‘discuss’ or ‘consider’ are weaker recommendations.
What is the evidence for treating after one attack?
The British Rheumatological Society guidelines in 2017 recommended that we offer urate-lowering treatment (ULT) after one attack. This was controversial. Its rationale was based on Delphi consensus process (expert and patient opinion) and included the following points:
- Chronic crystal deposition can be seen in multiple peripheral joint and peri-articular sites at the first attack of gout, and likely predates symptoms by some years.
- These intra-articular crystals lead to ongoing joint damage and chronic arthritis.
- 62% of patients with gout will have a second attack within 12m. Risk increases proportionally with SUA level, and, at SUA >500micromol/l, the risk is almost 100%.
There is an absence of more robust evidence to support this stand (DTB 2018;56(1):9).
NICE looked again at the evidence and found only 3 studies that were of high enough quality to consider.
Based on this evidence, NICE recommends “discussing and considering ULT for ALL people having a first or subsequent gout flare”, which largely aligns with the BSR guideline.
NICE also pointed out that delaying ULT initiation beyond the first attack may represent a missed opportunity.
Treat-to-target approach
Should we be treating to target? Yes
NICE looked at 2 RCTs comparing treat to target with usual care (continuation of current dose or no treatment), and found that treat to target:
- Increases flare frequency at 1 year.
- Reduces flare frequency at 2 years.
NICE concluded that treat-to-target approach was justified based on cost-effectiveness, flare frequency and quality-of-life outcomes.
What targets has NICE chosen?
There is an absence of evidence as to the best SUA level when using a treat-to-target approach.
NICE recommends ≤360micromol/l for most people but ≤300micromol/l for some – why?
- Below 360micromol/l, monosodium urate crystal deposition stops, and instead crystals start to dissolve.
- While a lower SUA target helps crystals dissolve more quickly, achieving 360micromol/l is easier and cheaper than the more stringent 300micromol/l target.
NICE therefore settled on 360micromol/l for most, and 300micromol/l for those with tophi, chronic gouty arthritis or who are still having flares when treated to SUA <360micromol/l.
Monitoring once at target
Once stable, an annual SUA check is required as it can increase with age, changes in lifestyle, comorbidities or medications. Further ULT uptitration may be required.
Can we overtreat gout?
The EULAR guidelines suggested that, in the long-term, SUA should not be <180micromol/l (3mg/dL). Why? It’s thought that uric acid may protect against some neurodegenerative conditions (Ann Rheum Dis 2017;76:29).
The FAST trial, discussed below, did not demonstrate any adverse outcomes resulting from low urate levels.
NICE does not warn against overtreatment in the 2022 guideline.
Choice of urate-lowering therapy agent
In contrast to the BSR guideline, NICE does not favour allopurinol over febuxostat as 1 year of treatment had minimal cost differences. Febuxostat is easier to titrate than allopurinol, and more likely to achieve SUA target. It is associated with more flares than allopurinol.
What about people with cardiovascular disease?
Due to the MHRA recommendation (MHRA, 17 July 2019) that febuxostat should be avoided in those with pre-existing major cardiovascular disease (unless no other therapy options are appropriate), NICE suggested that allopurinol should remain first-line treatment in this group. NICE’s recommendation remains unchanged following a update from the MHRA (MHRA, 25 May 2023). There’s more information about febuxostat and cardiovascular risk below.
Flare prophylaxis during ULT initiation
- Surprisingly, NICE gave the impression that prescribing a prophylactic drug to prevent a flare is optional: it used the word ‘discuss’ rather than its usual ‘offer’ (the BSR guideline recommended always using prophylaxis).
- NICE recognised that ULT could be started during an acute flare if absolutely necessary, e.g. frequent flares.
If using prophylaxis, NICE recommends:
| First line | Second line (for those where colchicine is unsuitable) |
| Colchicine |
NSAID Corticosteroid (off label) |
- IL-1 inhibitor (canakinmab) was not recommended by NICE (insufficient evidence).
Why?
This was because there were 3 studies looking at colchicine and none for NSAIDs in this situation.
Flare management while on ULT
- Do not stop the urate-lowering therapy!
- If already taking a prophylactic dose of a flare treatment, e.g. low-dose NSAID, increase to the higher dose.
- If on long-term maintenance ULT (i.e. no colchicine, NSAID or oral corticosteroid), simply add-on the acute flare-management medication until the flare settles. Is your patient treated to target?
How long should we use prophylaxis for?
Trying to find a simple answer is like trying to unscramble an egg!
If you just want a quick answer – see the table!
- NICE doesn’t suggest a dose or duration of prophylaxis; it simply states, “offer colchicine while the target serum urate level is being reached”. That’s not very helpful.
- Next, we turn to the trusty BNF…which is more helpful. In fact, it mandates flare prophylaxis for febuxostat: “Administer prophylactic NSAID or colchicine for at least 6 months after starting febuxostat”. That’s quite prescriptive, in contrast with NICE which seems quite relaxed about the whole issue!
We have an answer for febuxostat, but what about allopurinol?
- The BNF is disappointing. It simply says that “initiation or up-titration of treatment may precipitate an acute attack of gout”.
- OK, so what about the excellent BNF treatment summary for gout? Here we find NICE’s statement duplicated: “Colchicine should be offered as prophylaxis while the target serum urate level is being reached”.
I feel like I’m chasing my tail!
- What does the older BSR guideline say? This is more helpful and suggests: “colchicine 500 micrograms bd or od should be considered as prophylaxis against acute attacks resulting from initiation or up-titration of any ULT and continued for up to 6 months. In patients who cannot tolerate colchicine, a low-dose NSAID or coxib, with gastroprotection, can be used as an alternative providing there are no contraindications.”
At last! An answer…
| Drug | First-line prophylaxis |
| Allopurinol | Up to 6 months with colchicine 500 micrograms once or twice daily (thank you, BSR) |
| Febuxostat | At least 6 months with NSAID or colchicine (dose not specified, BNF) |
Febuxostat and cardiovascular disease
Does febuxostat cause increased death from cardiovascular causes? This is an interesting and evolving area of evidence where there’s still some uncertainty. Here, we summarise the key trials:
The CARES trial
- In 2018, the CARES trial demonstrated an association between febuxostat and increased cardiovascular and all-cause mortality in patients with gout and major cardiovascular disease (the all-cause mortality was raised because of the increase in cardiovascular deaths) (NEJM 2018;378:1200). The CARES trial was not designed to establish causality; this was an association.
- Subsequently, the MHRA issued a warning to avoid using febuxostat in those with pre-existing major cardiovascular disease, unless there were no other options (MHRA, 17 July 2019).
The FAST trial
- In 2020, evidence from the European FAST trial appeared to contradict the CARES trial (Lancet 2020;396:1745).
- It concluded, “Febuxostat is non-inferior to allopurinol therapy with respect to the primary cardiovascular endpoint, and its long-term use is not associated with an increased risk of death or serious adverse events compared with allopurinol”.
Why the difference?
Both studies were well designed, so why the difference?
- The populations were very different. ALL participants in the CARES study had pre-existing major cardiovascular disease (compared with about a third in the FAST trial).
- The median age was 64–65y in the CARES study, and 71y in the FAST trial.
- Those in the FAST trial were already established on allopurinol prior to inclusion, while the CARES trial started with people on no urate-lowering therapy.
- There was a lead-in phase in the FAST trial which may have resulted in a lower dropout rate than seen in the CARES trial.
There is still some uncertainty. The FAST trial findings were reassuring, but only about a third in that trial had cardiovascular disease (and they’re the ones we’re interested in).
After appraisal of the available evidence, NICE (in 2022) made its recommendation to use allopurinol first line in those with known ‘major’ cardiovascular disease, and hinted the regulators may be considering further evidence on the subject.
It turns out that NICE was correct. In May 2023, the MHRA updated its warning to reflect evidence from the European FAST trial. The warning changed from “avoid treatment with febuxostat in patients with pre-existing major cardiovascular disease” (MHRA, July 2019) to “caution is required if prescribing febuxostat in patients with pre-existing major cardiovascular disease” (MHRA, May 2023).
For individuals with tophi or those initiating urate-lowering therapy, the MHRA urged greater caution. It suggested that the drug dose should be “titrated appropriately to minimise gout flares’ and thus avoid “additional inflammation” – the inference being that this may have negative consequences in those with pre-existing cardiovascular disease.
The MHRA goes on to remind us that NICE states that allopurinol should be offered first line to those with major cardiovascular disease.
Drug doses
NICE does not specify drug doses for management of either the acute flare or for long-term urate-lowering therapy.
In fact, NICE didn’t specify the drug name in some situations, choosing to use a very generic ‘an NSAID’ or ‘low-dose oral corticosteroid’. NICE doesn’t give an explanation, but we think this is likely to be due to the absence of evidence to prove superiority of one NSAID over another, or that prednisolone is any better than dexamethasone in this situation.
We suggest, as always, that you check the BNF, but we summarise some suggestions taken from the BNF and based on usual UK practice in the following table.
| Flare treatment and prophylaxis (during urate-lowering therapy initiation) | |
| Naproxen | Flare: 750mg then 250mg every 8 hours until attack has passed (+/- PPI cover). |
| Prophylaxis: low-dose 250mg twice daily (off licence) (+/- PPI cover). | |
| Colchicine | Flare: 500micrograms 2–4 times daily until symptoms relieved, max 6mg in acute course. |
| Prophylaxis: 500micrograms once or twice daily (caution if eGFR <60). | |
| Prednisolone | Flare: 30–35mg once daily for 3–5 days (off licence). |
|
Prophylaxis:
We were unable to find a specific dose recommendation for this situation, and suspect most of us would not use prednisolone in this situation. | |
| Intra-articular steroid for acute flare |
|
| Intra-muscular steroid | Flare: triamcinolone 40–60mg (off licence). |
|
Prophylaxis:
| |
| Urate-lowering therapy | |
| Allopurinol |
Check baseline renal function (see below for adjusted doses). Rashes are common but a rare exfoliative reaction may occur (Stevens-Johnson syndrome or toxic epidermal necrolysis). Advise your patient to stop allopurinol and seek advice should a rash develop. The 2017 BSR guideline recommended the following:
Do not stop allopurinol in the event of a flare. Lifelong treatment. |
| Febuxostat |
Check baseline LFT. A prior hypersensitivity rash with allopurinol may occur with febuxostat. 80mg once daily, uptitrated after 2–4 weeks to 120mg once daily if SUA target not met. Do not stop febuxostat in the event of a flare. Lifelong treatment. |
Doses of allopurinol in impaired renal function
A DTB review on the management of gout included a helpful table of recommended starting doses of allopurinol in renal impairment. In these patients, serum uric acid and renal function should be monitored every 3m in the first year, then annually. Advice from renal physicians may be needed for dose escalation (DTB 2018;56(1):9).
eGFR (ml/min/1.73m2) |
Allopurinol starting dose |
<5 |
50mg weekly* |
5–15 |
50mg twice weekly* |
16–30 |
50mg every 2d* |
31–45 |
50mg daily* |
46–60 |
50mg and 100mg alternate days |
61–90 |
100mg daily |
*NICE recommends specialist referral for management of those with gout if CKD 3b or worse, i.e. eGFR <45.
Antihypertensives and gout
Which is the best antihypertensive for patients with gout?
A UK-based study of over 24 000 patients with gout used the CPRD to answer this question (BMJ 2012;344:d8190). It looked at the relative risk of gout associated with the use of different antihypertensive drugs:
| Antihypertensives that lower gout attack risk | Antihypertensives that increase gout attack risk |
|
Calcium channel blockers Losartan |
Diuretics Beta-blockers ACE inhibitors Non-losartan ARBs |
Something to consider when you are reviewing your hypertensive patients with gout, particularly if they are having recurrent attacks?
Is urate an independent risk factor for cardiovascular disease?
Patients diagnosed with gout are at increased risk of metabolic syndrome. There is an absence of evidence that urate-lowering treatment alters that risk in a meaningful way (BMJ 2017;357:j2376).
- Meta-analyses have shown that urate-lowering treatment has marginal benefits in improving surrogate markers of cardiovascular disease, e.g. blood pressure, endothelial function and renal function.
-
There have never been any studies that have shown that urate-lowering treatment reduces the risk of important endpoints like heart attack, stroke and mortality.
- The ALL-HEART trial, a UK-based, prospective RCT, enrolled those aged >60y with established cardiovascular disease but without a history of gout. It randomised just under 6000 participants: half received 600mg allopurinol (300mg if coexisting CKD) and the others got ‘usual care’. The primary outcome was a composite of ‘non-fatal MI or non-fatal stroke or cardiovascular death’. There was no difference in the outcome between the two groups (Lancet 2022;400:1195). This was a large, well-conducted RCT with good, solid endpoints, performed in an ‘at-risk’ population. As such, it appears to be a good piece of evidence disproving the hypothesis that allopurinol may help prevent cardiovascular disease.
What does this mean in practice?
This means that we should manage the cardiovascular risk as for any other patient, with lifestyle interventions, management of hypertension and lipids, etc.
We should also discuss the advantages and disadvantages of long-term urate-lowering treatment, and be honest about the current limitations of the evidence.
How should we organise gout care?
Nurse-led (or pharmacist-led) clinics. Probably.
Gout is a long-term condition. In keeping with other long-term conditions, a UK-based RCT study demonstrated that nurse-led care can be more clinically-effective and cost-effective than GP-led care (Lancet 2018;392;1403).
More than 500 patients with ongoing gout were assigned to receive nurse-led care or ‘usual’ GP-led care. The nurses were research nurses who had been trained in the management of gout. They spent more time on patient education and individualising explanations, and in engaging patients in shared decision-making (i.e. recommended best practice). This study had the highest retention rates of any gout trial (at 2y, 91% of the nurse-led group), almost certainly reflecting the benefits of full information and positive patient–practitioner relationship and patient engagement. There will be a follow-up study implementing the same protocol for practice nurses.
Patients were followed-up for 2 years.
Outcome measures at 2 years |
Nurse-led care (% of patients achieving outcome) |
Usual care (% of patients achieving outcome) |
Serum urate <360micromol/l |
95% |
30% |
Serum urate <300micromol/l |
88% |
17% |
Taking urate-lowering therapy |
96% |
56% |
Two or more flares |
8% |
24% |
Four or more flares |
1% |
12% |
Presence of tophi |
3% |
11% |
Mean dose of allopurinol |
460mg/day |
230mg/day |
Mean number of contacts over 2 years |
9 face to face 8 telephone (mostly in the first 6 months) |
<1 face to face |
Note that in the nurse-led group, the number of flares was greater in the first 3 months of year 1, but lower than the usual care group after the first 6 months, likely reflecting uptitration of urate-lowering therapy. Allopurinol was successful and well tolerated even in the 20% with CKD stage 3.
Cost-effectiveness analysis suggests that at year 2, it would cost £5066 per QALY gained, modelled to be £286 by year 3 (so, a big upfront cost, but well below the NICE threshold of £20 000/QALY), with the potential to save costs by year 5.
Similar, though smaller, benefits have been seen in smaller US studies of pharmacist-led care.
So, nurse-led care led to better patient outcomes and is cost-effective, though more expensive for us to administer in primary care. Longer-term follow-up will tell us if this initial investment is offset by later financial savings.
|
Gout
|
|
|
Audit your care of gout patients – search for all gout patients on your list.
|
|
|
Useful resources: Websites (all resources are hyperlinked for ease of use in Red Whale Knowledge)
Useful resources for patients:
|
This information is for use by clinicians for individual educational purposes, and should be used only within the context of the scope of your personal practice. It should not be shared or used for commercial purposes. If you wish to use our content for group or commercial purposes, you must contact us at sales@red-whale.co.uk to discuss licensing, otherwise you may be infringing our intellectual property rights.
Although we make reasonable efforts to update and check the information in our content is accurate at the date of publication or presentation, we make no representations, warranties or guarantees, whether express or implied, that the information in our products is accurate, complete or up to date.
This content is, of necessity, of a brief and general nature, and this should not replace your own good clinical judgment or be regarded as a substitute for taking professional advice in appropriate circumstances. In particular, check drug doses, side effects and interactions with the British National Formulary. Save insofar as any such liability cannot be excluded at law, we do not accept any liability for loss of any type caused by reliance on the information in these pages.
Here is the link to our terms of use.